Metal corrosion is universal in the nature and the corrosion prevention for metals plays an important role everywhere in national economic development and daily life. Wondering what causes the interior of a water tank to corrode? Want to know how cathodic protection works? You’re just 54 seconds away from finding out from our President, Jim Skilton.
Translation
Cathodic Protection inside your water tank right?
In the old days you used to hang rods of zinc and aluminium inside, they are less noble metal than the steel so if corrosion has gonna happen,
it’s gonna go after those rods instead of the steel, now how you use cathodic protection, you put electric charge in the water right?
so where is your cathodic protection? where ever the water touches the steel right! so where
I’m getting at is, where is that you’re gonna have premature failure is in the roof and cathodic protection doesn’t protect the roof.
Only protection is where the water touches the steel, that’s the only protection. So my point is your worst place that you’re
gonna have corrosion is your roof and it doesn’t do anything to stop.
Learn More about Our Interior Paint Coatings Here.
Below is the Image of Simple impressed current system. A source of DC electric current is used to help drive the protective electro-chemical reaction.
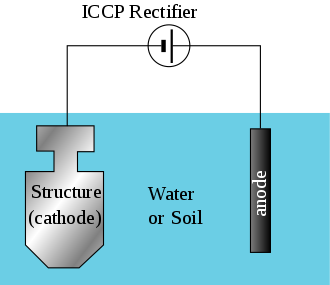
In a paper written by NAVFAC LANT (Naval Facilities Engineering Command Facilities) a division of engineers in the United States Navy named CATHODIC PROTECTION SYSTEM FOR STEEL WATER TANKS we can see how the different type of tests and execution of the system takes place. The paper published in October 2012 provides a great insight on how a traditional system for Water Tanks Manitenance and Water Tower Maintenance for an overall well being of water tanks takes place.